 
The positive electrode is called the anode. The current is carried by streams of tiny particles given off by the negative electrode, called the cathode. We need not discuss in detail the various experiments performed with this apparatus we will only state the conclusions drawn from them.  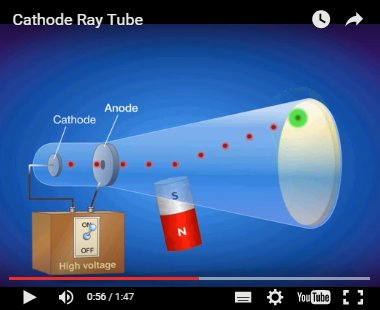
If the tube has been partially evacuated before sealing (that is, almost all the gas has been pumped out of it), the application of a high voltage from the power source across the two electrodes gives rise to a glow inside the tube, and simultaneously, a current begins to flow between the electrodes. If the tube is full of air or some other gas, no current flows between the electrodes, regardless of how large a voltage is applied from the power source. Within the tube are two metal plates called electrodes, which are connected to an outside power supply. This apparatus consists of a glass tube sealed at both ends. Matter in gas-discharge or cathode-ray tubes.įIGURE 4.1 Diagram of a cathode-ray tube.įigure 4.1 is a diagram of a cathode-ray tube. The experiments that proved its existence were studies of the properties of The electron was discoveredĪnd its properties defined during the last quarter of the nineteenth century. The atomic model usedīy chemists requires knowledge of only electrons, protons, and neutrons, soĪn electron is a tiny particle with a mass of 9.108 X 10 -28g andĪ negative charge. That atoms contain many different subatomic particles such as electrons, protons,Īnd neutrons, as well as mesons, neutrinos, and quarks. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
